Process-Level Studies Focused on Drought, Temperature and Neutron Imaging
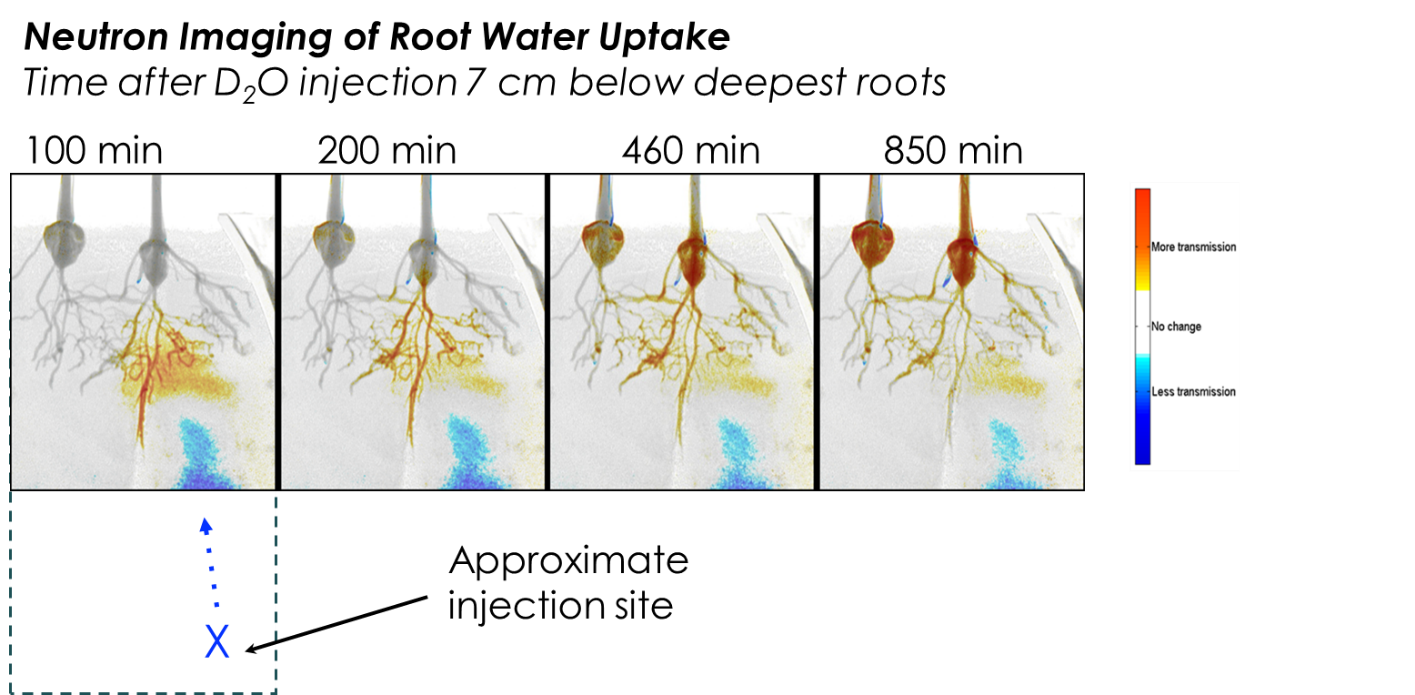
Assessment of root function in situ is extremely difficult, but advances in imaging technology are allowing unprecedented insight into root dynamics. Neutron imaging is highly sensitive to hydrogen ions, thus biological material and water are readily visible, which allows for in situ assessment of root structure, root growth, root water uptake & internal root water transport. In addition, near-root soil water dynamics can be assessed, which can be used to improve models of root water uptake and its linkages to root traits. Other key root processes that models are sensitive to include dynamics of root carbon allocation, relationships between roots and mycorrhizal fungi, root nutrient uptake, and root respiration. Here, we leverage neutron imaging techniques to assess in situ root water dynamics, and hyphal exclusion or root exclusion chambers to assess in situ respiration dynamics in response to environmental changes. In addition, root impacts on soil hydraulic properties are being assessed.
How do roots and hyphae affect soil hydraulic properties?
Earlier work using the HFIR neutron beamline at ORNL quantified soil water movement and root water uptake patterns in response to drought. Uptake was found to range from 0.003 to 0.012 g cm-2 h-1. Model simulations to recreate the dynamics using bulk soil hydraulic properties failed due to impact of roots and possibly mycorrhizal fungal hyphae on hydraulic conductivity, changes in pore space and hydraulic redistribution (Dhiman et al. 2018. Plant Soil 424:73-89).
Proposed work will test differences in bulk soil hydraulic conductivity and soil water release parameters using 1) Soil, 2) Soil + Mycorrhizal Hyphae, 3) Soil + Roots, 4) Soil + Roots + Mycorrhizal Hyphae
We will link hydraulic parameters to root and hyphal traits, assess spatial patterns of soil water release curves and root density using neutron imaging at ORNL, then model water extraction dynamics using the new parameters.
How do Root, Hyphal & Microbial Respiration respond to drought, temperature?
How does this vary by species?
Earlier work conducted during the PITS 13C carbon partitioning studies demonstrated that a large fraction of new photosynthate can be rapidly transferred belowground to roots, pass through the roots to mycorrhizal symbionts, and then be respired by the fungal hyphae. This pattern is demonstrated by the similar magnitude in surface 13CO2 efflux from combined root+fungal chambers, and fungal only chambers after a field labeling experiment in a dogwood stand (below)
Proposed work will test the seaonal patterns of root respiration rates in situ, with a specific goal to test different species and separate root from fungal respiration rates using custom-built chambers (e.g,. below left) that selectively exclude roots or hyphae. Example from a white oak mesocosm study subjected to weeks-long drought (below right; Ficken and Warren 2019. Plant and Soil 435: 407-422). Root/hyphal respiration work will leverage ongoing work at MOFLUX, SPRUCE, Morton Arboretum as well as the greenhouse and growth chamber facilities at ORNL. Our goals include improved understanding of the mechanistic linkages between carbon uptake, growth, carbon allocation belowground, and separation of belowground respiration from various sources, and how respiration rates respond to changes in environmental conditions.